|
Chemical Equation Expert - Free download and software reviews. Please describe the problem you have with this software. This information will be sent to our editors for review. Balancing Chemical Equations Main Concept A chemical equation describes the changes that occur during a chemical reaction. The equation shows the reactants on the. How do you know if a chemical equation is balanced? Balancing Chemical Equations. How do PhET simulations fit in my middle school program? Sarah Borenstein: MS. 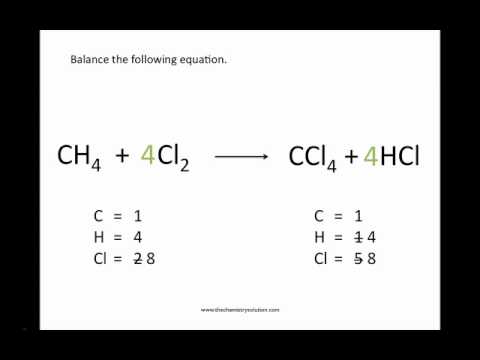
BBC - GCSE Bitesize: Chemical equations. You should be able to write word equations for the reactions you study in GCSE Science or GCSE Chemistry. If you are taking the Higher Tier, you should also be able to write and balance symbol equations. Balanced symbol equations show what happens to the different atoms in reactions. For example, copper and oxygen react together to make copper oxide. Take a look at this word equation for the reaction: copper + oxygen .
Copper oxide is the product because it is on the right of the arrow. If we just replace the words shown above by the correct chemical formulas, we will get an unbalanced equation, as shown here: Cu + O2 . To make things equal, you need to adjust the number of units of some of the substances until you get equal numbers of each type of atom on both sides. Here is the balanced symbol equation: 2. Cu + O2 . This matches what happens in the reaction. Two atoms of copper react with two atoms of oxygen to form two molecules of copper oxide. Remember: never change a formula to balance an equation.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
December 2016
Categories |
 RSS Feed
RSS Feed
